Veterinary Patient Multi-Parameter Monitor Medical Clinic Equipment Animal Use
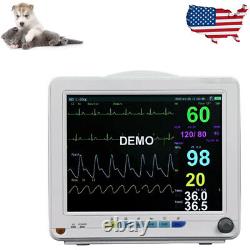
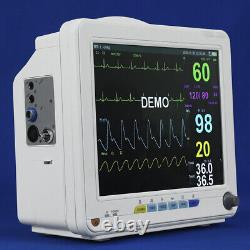
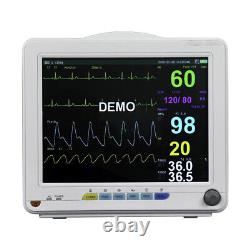
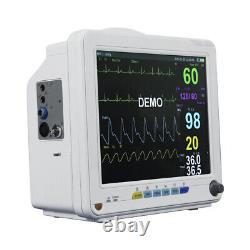





This monitor is suitable for felines, canines and other animals, may monitor the physical parameters, such as electrocardiograph (ECG), noninvasive blood pressure (NIBP), oxygen saturation (SpO2), respiration rate (Resp), body temperature (Temp) and so on, can display maximum 8 waveforms and all information of the parameters monitored in the same screen. Ergonomic design, Lightweight, compact and portable, convenient for use. 12 TFT display, configurable up to 8 waveforms, enables maximum data visibility for the most critical parameters.
Unique digital SPO2 technology can work accurate during motion and low perfusion. Rich screen layout, standard, large font, trend graph, OxyCRG, ECG full lead and other. Strong data storage: 720 hours trends, 10000 group NIBP, 1024 alarms events. ST analysis, arrhythmia analysis, pacemaker analysis, drug analysis and calculation.
Audible & visual alarm available, adjustable. Super long life rechargeable Lithium battery up to 4 hours of working time. This monitor provides the function of computation for 21 kinds of medicines and the titration table.
It is providing fast and reliable measurements for the veterinary patients. The measuring size of patients animals range from the small to big animals such as cats dogs, horses.
It is suitable for vet hospital, clinic, and animal labs. ECG, NIBP, SPO2, TEMP, RESP, PR, Rechargeable Lithium battery. Full lead: I, II, III, AVL, AVR, AVF, V. Gain: 5mm/mV, 10mm/mV, 15mm/mV, 20mm/mV and 25mm/mV.
Scanning speed: Changeable with three grades of 12.5mm/s, 25mm/s and 50mm/s. Band width: 0.05Hz-100Hz(3dB). Scope: 15 times/minute to 300 times/minutes. Variation of heart beat LORENZE drawing: Analysis on heart beat data for the current 5 minutes with histogram during RR period.Arrhythmia: Twenty kinds of common abnormal ECG can be analyzed, with ECG data of 1000 times of arrhythmia kept and with 10 seconds kept for each time. Accuracy:±1% 90%-100%;±2% 70%-89%.
Automatic detection period:1 minute to 240 minutes. Measurement Range:0 150 BrPM. Measure and Alarm Range:0 50. Operating Temp: 0 to 05°c. Storage Temp: -20 to +50°c. Relative Humidity: 15 to 95%(Operating), 15 to 95%(Storage). Power Supply:100240 VAC, 50/60 Hz, Pmax=90VA Fuse T1.5A. Statement:The sale of this item may be subject to regulation by the U. Food and Drug Administration and state and local regulatory agencies.The Fingertip Pulse Oximeter is certified with the US FDA 510K No. K070371, the CE & TUV of Eureope and it is on the Australian Register of Therapeutic Goods (ARTG) with the code 136606.
The Powered Surgical Instrument / Speed 808 System is certified with the US FDA 510(k) Number:K132989. The Powered Surgical Instrument / Hair Remove Device is certified with the US FDA 510(k) Number:K180353. The Powered Surgical Instrument / Hair Remove System is certified with the US FDA 510(k) Number:K141973. Massager, vacuum, light induced heating / Slimming Treatment Device is certified with the US FDA 510(k) Number:K161892. You pay us what you see on the invoice, i.Please make sure it is correct. Or replace item for you free of charge. This item is in the category "Business & Industrial\Healthcare, Lab & Dental\Medical & Lab Equipment, Devices\Patient Monitors". The seller is "xinchen02" and is located in this country: US.
This item can be shipped to United States.
- Brand: Unbranded
- Intended Use/Discipline: Emergency Medicine, Family Medicine, General Surgery
- Features: Audible & visual alarm
- Model: RPM-9000A
- Warranty: 12 month
- Display: 12\
- suitable for: vet hospital, clinic, animal labs,etc
- Standard configuration: ECG, NIBP, SPO2, TEMP, RESP, PR