CV Medical Nucart
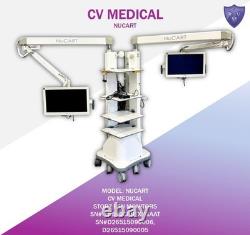













The equipment is not fully tested but appears to be operational. The equipment is clearly used and in good cosmetic condition. Equipment might have minor scratches, show fading, or include small dents or other blemishes.
Please carefully review the listing, photos, and notes for any specific limitations identified. Only includes what is shown in the photos. The sale of this item may be subject to regulation by the U. Food and Drug Administration and state and local regulatory agencies.
Reprocessing will be required before patient use. It is up to the purchaser to meet all requirements and regulation certification processes that deemed necessary per FDA or any other regulatory agencies that may be applicable. As with any piece of medical equipment, this unit needs to be inspected and calibrated before being put into use.